What Determines an Element's Emission Spectrum Apex
The emission spectrum of a chemical element or chemical compound is the spectrum of frequencies of electromagnetic radiation emitted due to an atom or molecule making a transition from a high energy state to a lower energy state. Each elements emission spectrum is unique.
Tm Series Energy Dispersive X Ray Spectrometer Element Series
In order to fully appreciate how this works.
. This is its atomic line emission spectra. Emission spectrum of a chemical element is the spectrum of frequencies emitted due to an atom making a transition from a high energy state to a lower energy state. The emission spectrum of a chemical element or chemical compound is the spectrum of frequencies of electromagnetic radiation emitted due to an electron making a transition from a high energy state to a lower energy state.
The emission spectra of an element is the spectrum of radiation emitted due to an atom or molecule absorbing energies and transitioning from a high energy state to a lower energy state. Other hydrogen atoms can have the electron excited into the n 4 shell. Atoms molecules or ions that absorb radiation are known to be in an excited state.
Different jumps appear as coloured bands of light. The number of times a wave vibrates or passes a fixed point per second. Add a small grating and capture the spectra of stars.
I know that every element has a different emission spectra but what determines the wavelengths present in a spectra of any given element. Emission spectrum has characteristic energy proportional to the frequency of the light. The electrons gained energy from the heat of a flame to go to a higher energy level then fall back to their original lower energy level Calculating the wavelength of an energy transition.
The number of protons B. Emission Spectrum of elements and compounds Element Description drawing or picture of spectrum Hydrogen Helium Neon Mercury 1. The diagram given in the question.
Every element in the periodic table has a distinct set of electron energy levels so for a given element only photons of specific energies can be emitted. Certain compounds and elements burn with distinctive colors. Explain why an element or ion that is placed in a flame high temperature or high voltage may show.
View Homework Help - Quiz. Spectra from CHEMISTRY Honors Che at Apex High. The photon energy of the emitted photon is equal to the energy difference between the two states.
Each elements emission spectrum is distinct because each element has a different set of electron energy levels. The energy of the emitted photon is equal to the energy difference between the two states. Each jump corresponds to a particular wavelength of light.
What determines an elements emission spectrum. The emission spectrum of a star is the spectrum of frequencies for emitted electromagnetic radiation during the transition of an atoms electrons from a high-energy state to a low-energy state. This video shows how to capture gas tube spectra in the classroom.
The Figure below shows the atomic emission spectrum of hydrogen. The electron in some hydrogen atoms may be excited into the n 2 level. When an atom absorbs energy its electrons jump to higher energy levels.
RSpec Explorer is an excellent companion to our poster. How Elements Produce Emission Spectra When elements are vapourised and heated their electrons jump in energy level. N2 n1 1.
But just in case we cared what the numbers were using Sals numbers gives you an average atomic mass of 8769 u for strontium while the official. The emission lines correspond to the differences between various pairs of the many energy levels. Based on the following figure and what you have learned about Niels Bohrs model of the atom account for the difference in energy between the lines.
The size of the sample O B. Then they jump back down again. Ive heard that it may have something to do with the electron energy levels but I dont know if that is correct.
Use it to capture gas tube and other spectra in real-time. The lines photons are emitted as electrons fall from higher energy orbitals to lower energies. ν wavenumber of the electromagnetic radiation.
The number of protons. The value 109677 cm -1 is known as Rydberg constant for hydrogen. How can the flame tests or emission spectra of element be used to determine the presence or identity of.
Elements Can Be Identified From Their Spectra Each element produces its own set of spectral lines. The emission spectrum shows the different colors of light that an element emits. RSpec Explorer is an affordable 395 desktop spectrometer for physics chemistry and astronomy educators.
And using the numbers that he did with numbers with a low amount of sig figs accomplished that. The mass of the nucleus O C. Well each element on the periodic table has a unique light fingerprint that can be read from a distance.
The characteristic spectrum of colors emitted by an atom. Different elements emit different emission spectra when they are excited because each type of element has a unique energy shell or energy level system. When the electrons fall back to their resting states they emit photons of light.
The EM wave with the shortest wavelength and the most dangerous EM wave. The electron energy levels O D. An atomic emission spectrum is the pattern of lines formed when light passes through a prism to separate it into the different frequencies of light it contains.
You can look at the spectra and identify which elements are present. Spectra Question 10 2 points What determines an elements emission spectrum. Use the equation for the energy E of light Ehc 2.
There are many possible electron transitions for each atom. Each element has its own unique atomic emission spectrum. Test your knowledge on Emission spectrum.
The emission spectrum of an unknown element contains two lines - one in the visible portion of the spectrum and the other ultraviolet. Therefore when you are measuring the emission spectrum of an element only certain wavelengths of light are allowed and the pattern that is produced is unique for that substance. ν 109677 1 n 1 2 1 n 2 2 Where n1 1234.
The general formula for the hydrogen emission spectrum is given by. There are many possible electron transitions for. Id like to know what causes the wavelengths of the emission spectra of different elements.
With Sals example we were only trying to estimate the atomic mass to determine what element we had.

Potentially Toxic Elements And Microplastics In Muscle Tissues Of Different Marine Species From The Persian Gulf Levels Associated Risks And Trophic Transfer Sciencedirect
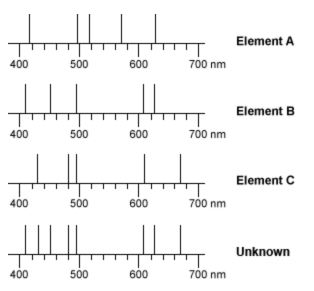
A Student Was Given An Unknown Sample Containing One Or More Elements The Line Spectrum Of Each Of The Possible Elements And The Spectrum Of His Unknown Sample Are Given Below Which
Improvement Of Te Polarized Emission In Type Ii Inaln Algan Algan Quantum Well

Stealth Aircraft Cuff Links Jet Fighter Cuff Links Stealth Aircraft Fighter Jets Cufflinks

Analytical Performance Of The Md Apgd Oes For Li Na Ca And Mg Download Scientific Diagram
Quantitative Modeling Of Near Field Interactions Incorporating Polaritonic And Electrostatic Effects
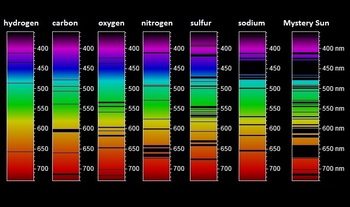
What Is An Emission Spectrum Example

The Electric Dipole Transition Matrix Element Of An Exciton As A Download Scientific Diagram

Sulfur Element Facts Properties Production Uses Study Chemistry Electron Configuration Ionization Energy
Why Is Each Element S Emission Spectrum Unique Quora

Ways To Improve The Eds Quantitative Results Accuracy Efficiency And Ezaf Scc Database
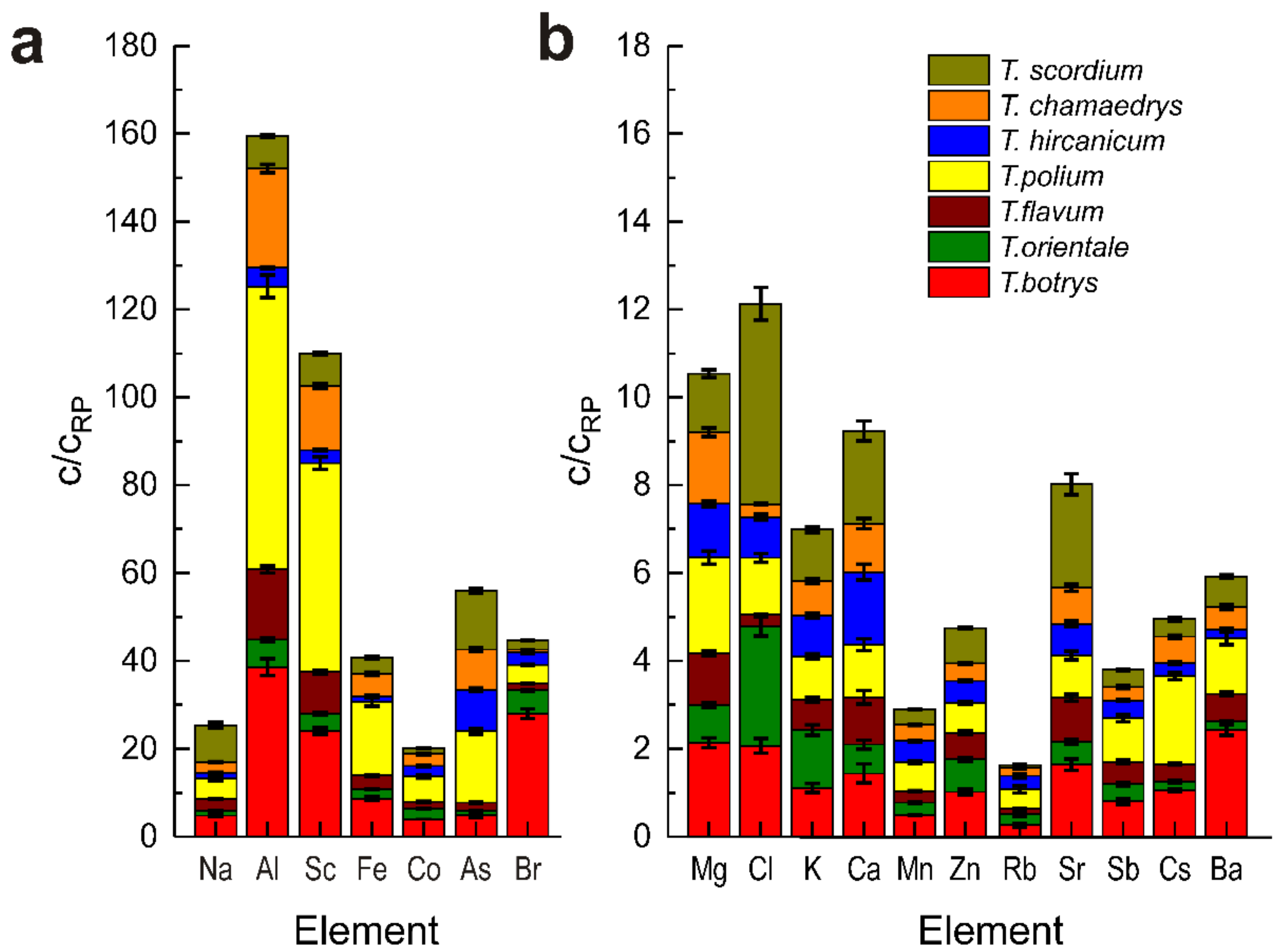
Agronomy Free Full Text Chemical Profile Elemental Composition And Antimicrobial Activity Of Plants Of The Teucrium Lamiaceae Genus Growing In Moldova Html

Growth And Xrd Elemental Mechanical Dielectric Optical And Photoconductivity And Surface Morphological Characterizations Of 2 4 Trifluoromethyl Phenyl 1h Benzimidazole Tfmphb Crystals For Electronic Mechanical Applications Springerlink

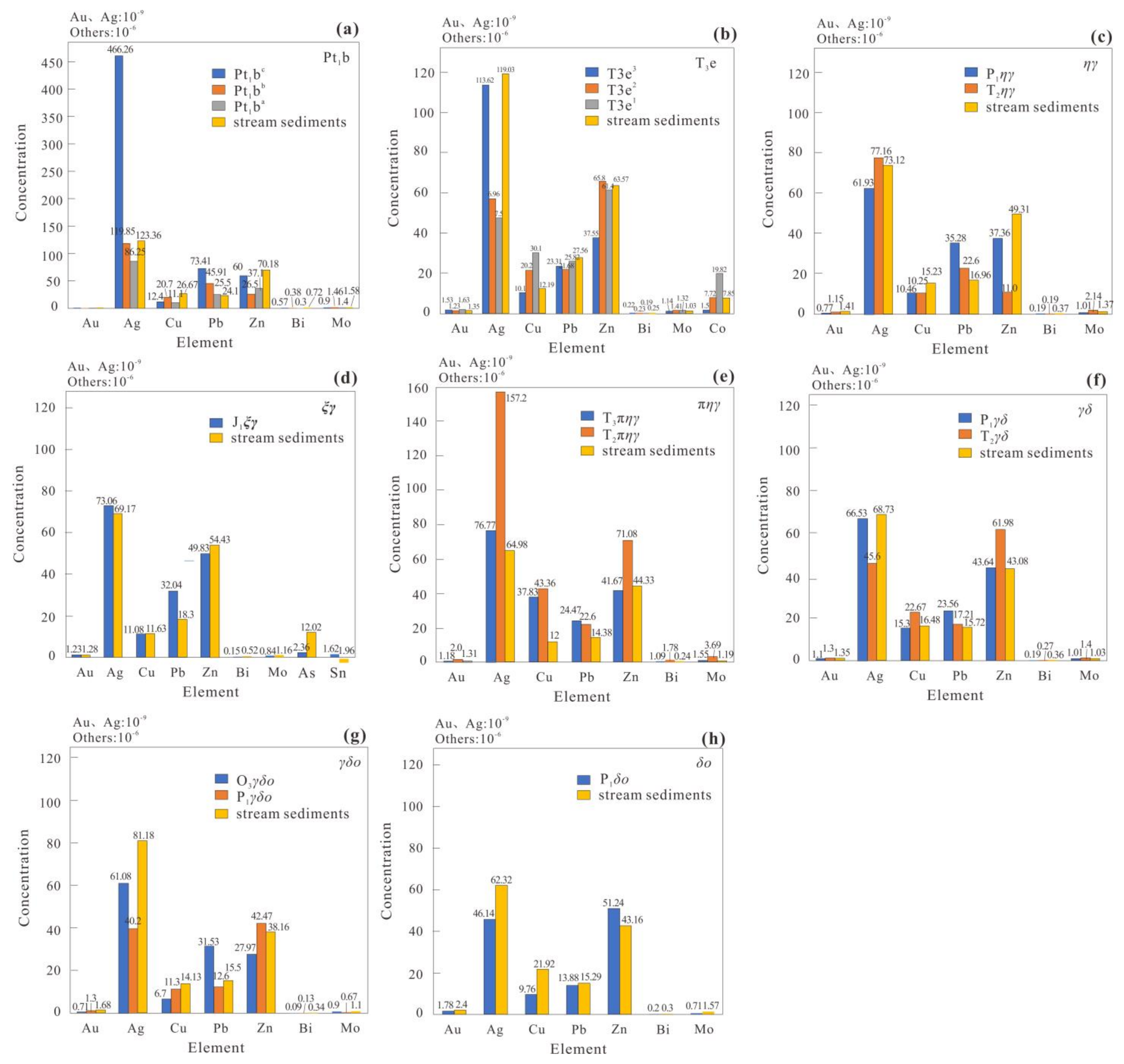
Applied Sciences Free Full Text Geochemical Association Rules Of Elements Mined Using Clustered Events Of Spatial Autocorrelation A Case Study In The Chahanwusu River Area Qinghai Province China Html

Antenna Separation An Overview Sciencedirect Topics

Antenna Separation An Overview Sciencedirect Topics

Antenna Separation An Overview Sciencedirect Topics

Pdf Simultaneous Polarization Filtering And Wavefront Shaping Enabled By Localized Polarization Selective Interference

A Hairpin Like Structure Within An Au Rich Mrna Destabilizing Element Regulates Trans Factor Binding Selectivity And Mrna Decay Kinetics Journal Of Biological Chemistry
Comments
Post a Comment